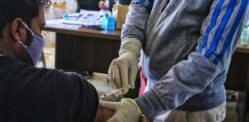
The trials involve volunteers over 18 years of age
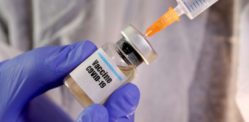
India’s first indigenous Covid-19 vaccine candidate Covaxin entered phase-3 testing on November 16, 2020.
Developed by Bharat Biotech in collaboration with Indian Council of Medical Research (ICMR), Covaxin is the front runner for vaccines being developed in India for Covid-19.
Krishna Ella, Chairman and Managing Director at Bharat Biotech, speaking virtually at a programme organised by the Indian School of Business said:
“We are working on another vaccine through nasal drops my feeling is by next year it will reach the population.”
He said that Bharat Biotech is the only vaccine company in the world which has a BSL3 production facility (Biosafety level 3).
In October 2020, the vaccine maker said it had successfully completed an interim analysis of Phase I and II trials of the vaccine and is initiating Phase-III trials in 26,000 participants.
On October 2, 2020, the firm sought the Drug Controller General of India’s (DCGI) permission to conduct phase-3 testing.
It was reported that the company will be conducting a randomised double-blind placebo-controlled multicentre trial of its Covid-19 vaccine.
In September 2020, Bharat Biotech said it entered into a licensing agreement with Washington University School of Medicine in St. Louis for a novel “chimp-adenovirus” (Chimpanzee adenovirus), which is a single-dose intranasal vaccine for Covid-19.
The trials began in Hyderabad with the first doses of the vaccine candidate being administered to volunteers at the Nizam’s Institute of Medical Sciences (NIMS).
The phase-3 trial will also be conducted in Andhra Pradesh at Guntur Medical College and at the King George Hospital in Visakhapatnam.
The trials involve volunteers over 18 years of age and they will be held at across 25 centres in the country.
According to the company, volunteers undergoing vaccination in the phase-3 trials will be monitored for Covid-19 over the next year.
Covaxin has already been monitored in 1000 subjects in phase-1 and 2 clinical trials and has shown promising safety and immunogenicity data.
As part of the Phase III study, volunteers will receive two intramuscular injections at an interval of around 28 days.
The participants will also be randomly assigned 1:1 to receive either two 6 mcg (microgram) injections of Covaxin or two shots of the placebo.
Being a double-blinded trial, neither the investigators, participants or the company will be aware of who is assigned to which group, the company said.
The news of advancement in India’s Covid-19 vaccine, Covaxin could not come sooner with cases rises tremendously on a daily basis and with the disease showing no end in sight.